![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
19 Cards in this Set
- Front
- Back
|
What are the four main different types of tissue? |
- Epithelial - Muscle - Connective - Nerve |
|
|
Connective tissue generally has two different categories in terms of shape and mechanical properties: |
- pulling and stretching - twisting and squashing |
|
|
Connective tissue is broadly divided into four categories: |
- Blood - Bone (compact and spongy) - Cartilage (hyaline, fibrocartilage, elastic) - Connective tissue proper (loose and dense) |
|
|
What are the different types of loose connective tissue proper: |
- Areolar - holds organs together, underlying epithelial tissues, blood vessels, nerves - Adipose - Reticular - composed of reticular fibres, found in the spleen, liver and lymph nodes |
|
|
What are the different types of dense connective tissue proper: |
- Regular - the collagen fibres run in the same diraction, such tssue has a very high tensile strength (tendon) - Irregular - less regularly arranged collagen fibres giving strength in all direction (dermis) - Elastic - in this tissue, elastin is the main fibre type; an example of this is found in the vocal fold |
|
|
What are the main characteristics of connective tissue? |
- Abundance of extracellular matrix - Cells are normally separated by ECM |
|
|
Examples of connective tissue in other phyla: |
- chitin exoskeleton in insects - main support - connective tissue layers surrounding many organs - CT attaching Malpighian tubules to the gut |
|
|
What are the functions of connective tissues? |
- Join the other tissues of the body - Mechanical support to organs - take the stress of movement - maintain shape - growth, development and repair |
|
|
What is the extracellular matrix? |
- A composite of insoluble fibres and soluble polymers |
|
|
What are the three principle components of the ECM? |
- Collagen - confer tensile strength - Proteoglycans - confer resiliency and compressibility - Matrix glycoproteins - elasticity, tissue cohesiveness & cell-ECM communication |
|
|
What are the features of collagen? |
- a family of ~29 fibrous proteins that share a common motif - trible helix - 3 polypeptides (α chains) intertwine to form a rod-shaped super-helix - type I collagen is the major collagen in CT |
|
|
What are the features of type I collagen? |
- high abundance of Glycine, Proline and Hydroxyproline - Gly-X-Y repeating motif - triple helix stabilised by hydrogen bonds - denatured collagen - gelatin |
|
|
What post-translational modifications does collagen go through: |
1) hydroxylation of proline (ascorbate is essential co-factor = keeps enzyme in active/reduced state) 2) hydroxylation of lysine 3) glycosylation 4) lysyl oxidase - initiation of collagen cross links |
|
|
What is the role of hydroxyproline residues? |
- stabilize the triple helix by increasing hydrogen bonds between α chains - more hydroxyproline = more hydrogen bonds = higher melting temperature |
|
|
How do α chains fold into a triple helix? |
- initiated by C-terminal propeptide - alignment - folds from the C terminal towards the N terminal |
|
|
Propeptides in the ECM are cleaved: |
- Converting soluble procollagen to insoluble collagen triple helical rods - cleavage of the C and N terminal propeptides - The cleavage is mediated by C and N proteinases |
|
|
How is type I collagen assembled (from molecules to fibrils)? |
- Three precursor chains assemble into triple helix with loose ends (procollagen) - procollagen peptidase = collagen - assembly of collagen into fibrils - adjacent molecules are displaced approximately 1/4 of their length = striated appearance seen in electron microscope - Collagen fibrils aggregate side-by-side in bundles into fibres |
|
|
How is type I collagen assembled (image)? |
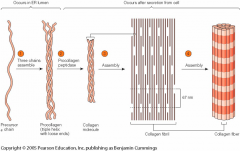
|
|
|
How does collagen cross-linking take place and what is the purpose of it? |
- initiated by oxidation of lysine or hydroxylysine residues by lysyl oxidase - stabilizes collagen fibrils |

