![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
360 Cards in this Set
- Front
- Back
|
Compartments ENT
|
-oral/oropharynx
-salivary gland sinonasal -nasopharynx -ear -larynx -skin -thyroid/parathyroid -lymph node -bone/soft tissue |
|
|
Oral/Oropharynx
|
Infectious
-candida Non-infectious;non-neoplastic: -extravasation mucocele -lingual thyroid -sarcoidosis Neoplastic: -squamous cell cacinoma -tonsillar lymphomas (heme) |
|
|
Candida sp.
|
Clin: Immunocompromised patients. "Thrush" = oral candidasis
Rad: Not done Path: Superficial infection by oral flora. Hyphal forms (spaghetti) and yeast forms (meatballs) |
|
|
Extravasation Mucocele
|
Clin: lower lip mass, floor of mouth cyst ("ranula")
Rad: Not done Path: Etio: minor salivary duct trauma Mucin extravasation (Alcian blue (+)) |
|
|
Lingual Thyroid
|
Clin:
-children-old age -dysphagia, foreign body sensation -10% are hypothyroid Rad: -scintigraphy to demonstrate other thyroid and function of lingual thyroid Path: -only thyroid gland in >70% -unencapsulated -low malignant potential |
|
|
Sarcoidosis
|
Clin: young adults with fever, weight loss, Pulmonary, cutaneous involvement
ENT: oral, S/N, larynx, salivary gland, ear RAD: Screen with CXR: hilar adenopahty ENT: non-destructive Path: Well-defined, non necrotizing granulomas -no vasculitis -elevated angiotensin-converting enzyme -lip biopsy (+) in ---> 50% |
|
|
Squamous Cell Carcinoma
|
Clin: Lower lip most common (sun, smoking)
tongue > FOM >gingiva > palate> tonsil (smoking) Rad: Staging: local extent, regional LNs, CXR Path: Etio: mutagens -prognosis a function of size, grade, stage |
|
|
Salivary glands
|
Infectious
-sialadenitis +/- sialolithiasis (stones) Non-infectious, non-neoplastic: -sjogren's syndrome Neoplastic: -pleiomorphic adenoma -mucoepidermoid CA -adenoid cystic CA |
|
|
Sialoadenitis +/- Sialolithiasis
|
Clin: inflammation, partoid or submandibular glands; Non-obstructive: staph, strep, mumps
Obstructive: duct stones +/- infection Rad: plain films to document stone (most sumbmandiulare are radio-opaque) Path: ductal stones: SMG>parotid -Acinar inflammation, fibrosis, atrophy |
|
|
Sjogren's syndrome
|
Clin: adults, F>M, associated with:
-keratoconjectivitis sicca -xerostomia -connective tissue disease (RA, SLE, PAN,etc) Rad: Not done Path: - major and minor glands involved -benign lymphoepithelial lesions -epimyopepithelial islands |
|
|
Pleiomorphic adenoma
|
Clin: Adults, F>M, with slow-grwoing, painless mass of major or minor salivary gland Parotid> others
Rad: Not done Path: -most common benign sal gl meoplasm -discrete, unifocal, encapsulated -epitheliu, myoepithelium, myxoid stroma -local recurrence potential, rare malignancy |
|
|
Mucoepidermoid carcinoma
|
Clin: children-adults, major or minor glands may have h/o prior irradiation
Rad: staging Path: most common malignant sal gl neoplasm -low/intermediate grade: mucous >epidermoid -high grade: epidermoid> mucous -mucicarmine stain (+) |
|
|
Adenoid cystic CA
|
Clin: adults, M+F, with salivary gland mass major>minor
RAD: ND Path: -unencapsulated, infiltrative, perineural -tubular or cribiform architecture -mucinous basement membrane material -course: local recurrences, late metastases |
|
|
Sinonasal
|
Infectious
-allergic fungal sinusitis Non infectious, non-neo -inflammatory (allergic) polyps -(sarcoidosi) -wegener's granulomatosis Neoplastic -schneiderian papillomas -(skin lesions of nassal vestibule) (derm) |
|
|
Allergic Fungal Sinusitis
|
Clin: Allergic resonse to fungi (aspergillus) Children-young adults with headache
Rad: sinus opacification, maxillary, ethmoid most common Path: mucoid plug with fungal hyphae (GMS Silver stain (+)) |
|
|
Inflammatory Polyps
|
Clin: any age, rhinorrhea, headaches, airborne allergens
Children: r/o CF and Kartagener's Rad: ST mass(es), in sinuses or nasal cavity Path: boggy polyps up to several cm -respiratory epith, stromal edema, eos |
|
|
Wegener's Granulomatosis
|
-Adults, M>F, ENT, lung and GU
ENT: nasal cavity>sinuses>other ENT Rad: sinus opacification, bone/ST destruction Path: -necrotizing granulomatous vasculitis - increase c-ANCA in >85% with active disease |
|
|
Schneiderian papillomas
|
-Adolescents, adults, local symptoms, ST mass on either nasal septum o lat wall
Rad: unilateral nasal cavity ST mass +/- sinus opacification Path: 3 histological types -fungiform/exophytic: septal wall, no increase risk of CA -oncocytic: lateral wall, 5-10% risk of CA -inverted: lateral wall, 5-10% risk of CA |
|
|
Nasopharynx
|
Infectious
-infectious mononucleosis Non-infectious, non-neoplastic -Sarcoidosis, Wegener's granulomatosis Neoplastic -teratoma -angiofibroma -nasopharyneal carcinoma |
|
|
Infectious Mononucleosis
|
Adolescents, young adults most common
-fever, sore throat, malaise -tender posterior LNs, hepatosplenomegaly Rad: none done Path: -tonsillitis/lymphadenitis -Reactive B and T lymphocyte hyperplasia -IgM and IgG serolgies against viral Ags -Self-limited in immunocompetent patients |
|
|
Inflammatory Polyps
|
Clin: any age, rhinorrhea, headaches, airborne allergens
Children: r/o CF and Kartagener's Rad: ST mass(es), in sinuses or nasal cavity Path: boggy polyps up to several cm -respiratory epith, stromal edema, eos |
|
|
Wegener's Granulomatosis
|
-Adults, M>F, ENT, lung and GU
ENT: nasal cavity>sinuses>other ENT Rad: sinus opacification, bone/ST destruction Path: -necrotizing granulomatous vasculitis - increase c-ANCA in >85% with active disease |
|
|
Schneiderian papillomas
|
-Adolescents, adults, local symptoms, ST mass on either nasal septum o lat wall
Rad: unilateral nasal cavity ST mass +/- sinus opacification Path: 3 histological types -fungiform/exophytic: septal wall, no increase risk of CA -oncocytic: lateral wall, 5-10% risk of CA -inverted: lateral wall, 5-10% risk of CA |
|
|
Nasopharynx
|
Infectious
-infectious mononucleosis Non-infectious, non-neoplastic -Sarcoidosis, Wegener's granulomatosis Neoplastic -teratoma -angiofibroma -nasopharyneal carcinoma |
|
|
Infectious Mononucleosis
|
Adolescents, young adults most common
-fever, sore throat, malaise -tender posterior LNs, hepatosplenomegaly Rad: none done Path: -tonsillitis/lymphadenitis -Reactive B and T lymphocyte hyperplasia -IgM and IgG serolgies against viral Ags -Self-limited in immunocompetent patients |
|
|
Teratoma
|
Clin: Majority in children, rare after age 2
-dysphagia or airway obstruction Rad: ST mas in NP Path: -germ cell tumor: 3 germ cell layers present -Mature vs. immature -pure teratoma or mixed with other GCTs -no malignant potential in children |
|
|
Angiofibroma
|
-adolescent--> young adult males
-nasal obstruction and epistaxis -lateral or posterior NP wall mass Rad: CT: ST mass with contrast enhancement Angiography: vascular hypertrophy Path -sessile, lobulated ST mass -benin fibrous tumor with increased numbers of thin-walled vessels without smooth muscle |
|
|
Nasopharyngeal carcinoma
|
-adults, M>W, China>US, associated with EBV
Rad: ST mass of lat NP (fossa of Rosenmuller) +/- bone destruction/invasion Path: -undifferentiated>squamous -undiff type has admixed benign lymphs |
|
|
Ear
|
Infectious
-ottis media Non-infectious, non-neoplastic -sarcoidosis Neoplastic -cholesteatoma -8th cranial nerve Schwannoma (neuro) -Paraganglioma -external canal squamous cell carcinoma |
|
|
Ottis Media
|
-young children with recent URI, fever, otalgia, decrease hearing, bulging immobile TM due to undrained middle ear fluid
Rad: Nlot done Path: never biopsied, Micro: sterile or (+) strep, staph, H. flu |
|
|
Cholesteatoma
|
Any age, peak 20s-30s, M>F, Hearing loss, pain, middle ear +/- mastoid mass
Rad: Not done Path: cyst of benign squamous epithelium with accumulation of loose keratin, may show foreing body reaction |
|
|
Jugulotympanic Paraganglioma
|
-Adults, F>M, middle ear ST mass associated with jugular vein bulb
Rad: CT:ST mass with local destruction Angio: well-vascularized Path: -benign but invasive paraganglioma -Chief and sustentacular cells, "zellballen" -synaptophysin/chromogranin AB stains |
|
|
External Ear/Ear Canal Squamous Cell Carcinoma
|
Adults, M>F
-non-healing mass or ulcer of external ear Rad: not done Path: Etio: sun exposure (actinic damage) Differential: melanoma, basal cell CA |
|
|
Larynx
|
Infectious
-squamous papillomas (HPV) -epiglottis Non-infectious, non-neoplastic -vocal cord polyps Neoplastic -squamous cell carcinoma |
|
|
Squamous Papillomas
|
Children: 2-3 yo, multiple lesions, frequent local recurrence
Adults: single lesions, less frequnet local recurrence, less frequent spread Rad: not done Path: -bland squamous papillomata -may have HPV changes -10% spread distally, 2%--> CA |
|
|
Actue Epiglottis
|
child or adult with fever, upper airway obstruction, stridor
Rad: Last view showing epiglottic edema Path: Acute inflammation, bacteria, usualy H. flu Course: may die if airways not estbalished |
|
|
Vocal cord polyps
|
adults with hoarseness and a history of voice abuse
-aka laryngeal nodule, "singer's nodule" Rad: Not done Path: -benign stromal expansion -benign surface squamous epithelium |
|
|
Squamous cel carcinoma
|
Adults, M>W
smokers/drinkers with voice changes RAd: for staging local extent, regional LNs, mets Path: Etio: tobacco, EtOH, prior neck irradiation Incidence: Glottic>supraglottic>subglottic |
|
|
Lung pathology
|
-airway diseases (non-infectious)
-infections (pneumonias) -carcinomas |
|
|
Normal Anatomy Anatomic compartments
|
-airways (trachea, bronchi, bronchioles)
-alveoli -peribronchial stroma -interstitium |
|
|
Airway Diseases
|
-Asthma
-Aspiration/aspiration pneumonia -emphysema |
|
|
Asthma
|
-airway hyper-responsivenes
-triggers: antigens, exercise, drugs, infections, stress -acute, usually reversible diffuse bronchial narrowing -wheezing, dyspnea Rad: alternating atelectasis and overexpansion Path: -edema, smooth muscle thickening, basement membrane thickening, mucous cell hyperplasia, increased submucosal eosinophils, thickened intralumenal mucous |
|
|
Aspiration
|
-children-foreign bodies
-adults: -gastric acid, food, foreign bodies (lipids in nasal drops) Rad: foreign object or resulting pneumonia ---> tpically RLL Path: -foreign material -->foreign body giant cell reaction with exogenous material |
|
|
Emphysema
|
-increased elastase activity
-cigarette smoking, inherited alpha 1-antitrypsin deficiency Rad: increased lung volumes, flat diaphragm Path: -dilation of distal airspaces with septal destruction |
|
|
Lung infections
|
-types of inflammatory response
-bacterial pneumonia an dcomplications -mycoplasma -viral -mycobacteria -fungal |
|
|
Types of inflammatory response
|
Neutrophils: usually bacterial and in alveoli
Lymphocytes: usually viral in interstitium/septae Granulomatous inflammation: usually mycobacterial or fungal |
|
|
Bacterial pneumonia
|
General concepts: patterns, disease progression
-pneumonococcus- the prototype -complications |
|
|
Bacterial Pneumonia- patterns
|
Bronchopneumonia
-scattered foci of consolidation in either a single lobe or multiple lobes -terminally ill patients -dependent regions of lung -common ultimate cause of death Lobar -complete consolidation of a lobe -encapsulated organisms |
|
|
Bacterial Pneumonia Agnets
|
Community acquired
-S.pneumoniae, Klebsiella, Hemophilus -things with capsules Hospital acquired "nosocomial" -drug resistant strains -methicillin resistant Staph. Aureaus (MRSA) Opportunistic infections -immunocompromised patients |
|
|
Bacterial pneumonia- Agents
Pathogenesis |
-Most bacteria are normal inhabitants of the nasopharynx or oropharynx
-reach alveoli by: aspiration (most), inhalation, hematogenous seeding, direct spread from adjacent site (rare) |
|
|
Pneumococcal pneumonia
|
-Streptococcus pneumoniae
-the prototype of bacterial pneumonia -encapsulated gram + cocci (diplococcus) -normal resident of the nasopharynx -often preceded by a viral infection, may alter bronchial secretions |
|
|
Pneumococcal pneumonia Pathology
|
Early: pulmonary edema and proliferation of bacteria, intra-alveolar accumulation of neutrophils and erythrocytes-so called "red hepatization"
Later: serum and fibrinous exudates, intra-alveolar organization, macrophages--- so called "gray hepatization" |
|
|
complication of bacterial pneumonia
|
-abscess
-pyothorax or epmyema -bacteremia |
|
|
Abscess
|
-walled off area of infection with destruction of pulmonary parenchyma
-often anaerobic oral bacteria -more common on right than left lung -fever, cough, foul smelling sputum -mortality 5-10% |
|
|
Pyothorax/empyema
|
-purulent material within the pleural space
-may become loculated, which requires drainage as well as antibiotics to treat |
|
|
Bacteremia
|
-25%
-bacteria in the bloodstream -endocarditis, meningitis, pericarditis |
|
|
Mycoplasma
|
-Mycoplasma pneumoniae
-lack cell wall, slow growing -culture very difficult, serum antibody testing to Dx Acute pneumonia and tracheo-bronchitis -milder than usual bacterial pneumonia -"walking pneumonia" -15-20% of pneumonias in developed countries Highly transmissible through airborne droplets Lymphocytic infiltrate, usually of the lower lobes |
|
|
Viral pneumonias General
|
-Immune compromised (HIV, organ transplants)
-infants may get CMV -lymphocytes in the interstitium -distinct cytologic inclusions for some viral infections |
|
|
Tuberculosis
|
-mycobacterium tuberculosis
-small, acid fast bacillus -slow growing, 3-6 weeks to culture -decrease in 20th century due to anti-TB drugs, re-emerging as a result of HIV and drug resistant strains -Primary and secondary forms |
|
|
Primary tuberculosis
|
-infection by inhalation of aerosolized droplets containing organisms
-caseous granulomatous inflammation results Ghon complex forms: -peripheral focus of infection (Ghon focus, often adjacent to iterlobar fissure) -infected lymph node (hilum or mediastinum) -90% of primary exposure asymptomatic |
|
|
Secondary tuberculosis
|
-reactivation of primary tuberculosis OR new infection in a previously sensitized patient
-caseating granulomas again: apex and posterior segments of upper lobes -may erode into a bronchus, creating a cavity: often in apex of lung, future home for aspergillus |
|
|
Complications of tuberculosis
|
Miliary TB
-multiple small granulomas in many organs -results from hematogenous dissemination -most any organ can be involved Hemoptysis -inflammatory response erodes into pulmonary artery Aspergilloma in cavities |
|
|
Fungal pneumonia general
|
-fungi are ubiquitous in soil and air
-most exposure fails to produce infection: body temperature arrests growth, phagocytosis by neutrophils and macrophages -risks: chemo, steriods, T-cell deficiencies Usually granulomatous inflammation Silver stains demonstrate organisms |
|
|
Aspergillosis
|
Several species (A.niger, A.fumigatus)
-septate hyphae with acute angle branching -live in soil and decaying plant material Causes several distinct types of disease -invasive aspergillosis -aspergilloma -allergic aspergillosis |
|
|
Invasive Aspergillosis
|
-organisms invade blood vessels
-infarction of lung, dissemination, exsanguination -Immunocompromised |
|
|
Mycetoma (fugus ball)
|
-within preexisting cavity, often TB cavity
-tangled mat of hyphae, non-invasive |
|
|
Allergic Aspergillosis
|
susceptible patients develop immunologic reaction to Aspergillus, esp. asthma patients
|
|
|
Lung Infection Points
|
3 Basic inflammatory responses: neutrophils, lymphocytes, granuloma
Most agnets have a characteristic resonse -bacteria: neutrophils -Viral/mycoplasma: lymphocytes -mycobacteria/fungus: granuloma A few infections are unique -tuberculosis: primary vs secondary -aspergillosis: 3 different lung diseases |
|
|
Lung Carcinoma
|
-malignant epithelial neoplasms
-squamous cell carcinoma -adenocarcinoma -small cell carcinoma |
|
|
Lung Carcinoma: Epidemiology
|
-expected incidence (2010): 222,000 (US)
-expected mortality (2010): 157,000 (US) ->85% of lung carcinoma deaths (and 30% of all cancer deaths) occur in cigarette smokers -risk is proportional to # of cigarettes smoked, 15-30x in heavy smokers -risk decreases with cessation of cigarettesmoking: baseline after 15 years |
|
|
Tobacco: Morbidity and Mortality
|
-premature atherosclerosis: major risk factor
-emphysema: linear with exposure 7% per 10years -chronic bronchitis -carcinoma of pharynx, larynx, lung, esophagus, bladder, kidney |
|
|
Squamous cell carcinoma
|
-smokers (98%)
-20-20% of common carcinomas Rad: central >> peripheral Path: squamous differentiation -desmosomes (intercellular bridges) -keratin production, e.g. ker pearls |
|
|
Adenocarcinoma
|
-30-40% of common carcinomas
-#1 lung carcinoma in non-smokers -80% of adenoCA's ocur in smokers Rad: peripheral > central Path: -gland formation -mucin production |
|
|
Small cell carcinoma
|
-20% of carcinomas
-smokers -commonly high stage at presentation -generally not treated with surgery -responsive to chemo/RT, but low 5 yr survival Rad: Central in >90%, frequent metastases to LNs and distant sites Path: -malignant cytology with nuclear molding -no nucleoli, little cytoplasm -high mitotic activity and necrosis |
|
|
Lung carcinoma Points
|
-3 major types: squamous, adeno, small cell
-most related to smoking: all types -most common in non-smoker: adenocarcinoma -central location: squamous, small cell -peripheral location: adenocarcinoma -treat with surery: squamous, adenocarcinoma -no surgery: small cell carcinoma |
|
|
Mass Lesions
(Summary) |
The cranial cavity is a closed box. Thus, the formation of a mass within the intracranial space requires that something exit from the intracranial space. CSF and blood are the first to exit, followed by shifting of parts of the brain. Such shifts or "herniations," cause serious brain dysfunction and eventuate in death if the mass effect is not reduced. Because of this potential for mass effect, some disease processes, which are relatively benign in other locations, may be lethal when located intracranially.
|
|
|
Mass Effect
|
The consequence of an intracranial mass (neoplasm, hematoma, abscess, edematous infarct, etc)
Mass may cause horizontal (left-right) brain shift and may cause downward herniation (Transtentorial & Tonsillar herniation) |
|
|
Transtentorial (uncal) hernation
|
Downward shift of medial temporal lobe & uncus thorugh tentorial incisure.
Due to supratentorial mass |
|
|
Tonsillar herniation
|
Downward shift of cerebellar tonsils through foramen magnum.
Due to posterior fossa mass |
|
|
Mass effect leads to intracranial pressure
|
Increased ICP causes headache, nausea, vomiting, pepilledema
Increased ICP may cause decreased cerebral blood flow, as intracranial pressure approaches or exceeds mean arterial pressure. Decreased cerebral blood flow leads to brain ischemia, which can cause irreversible injury |
|
|
Transtentorial (uncal) hernation
(Clinical Manifestations) |
Compression of 3rd cranial nerve causes DILATED PUPIL on side of lesion
Compression of cerebral peduncle causes CONTRALATERAL HEMIPLEGIA Distortion & downward displacement of midbrain causes abnormal respirations, coma, HEMORRHAGES IN MIDBRAIN with irreversible brainstem damage, and DEATH |
|
|
Tonsillar herniation
(Clinical Manifestations) |
Compression of medulla causes apnea & DEATH
|
|
|
Scalp Wound
|
INFECTION of a scalp wound may spread to the underlying skull (osteomyelitis), meninges (meningitis), or brain (brain abscess)
|
|
|
Skull Fracture
|
Indicates significant head trauma, but not necessarily brain injury
May be calvarial (skull cap) fracture or basilar (base of skull fracture) |
|
|
Complications of Calvarial (Skull Cap) Fracture
|
Tearing of dura may lead to tearing of its middle meningeal artery, which in turn leads to an ACUTE EPIDURAL HEMATOMA
Depressed skull fragments may cause BRAIN CONTUSION (bruising of brain) |
|
|
Complications of Basilar (Base of Skull) Fracture
|
Tearing of the meninges may lead to a CSF leak from ear (CSF otorrhea) or nose (CSF rhinorrhea), which predisposes to meningitis
Tearing of cranial nerves or stalk of pituitary gland |
|
|
Focal Intracranial Lesions
|
Head trauma, if significant, may cause focal intracranial lesions These focal traumatic lesions may result in localized neurologic deficits or a mass effect
May include: Contusion, Acute epideral hematoma, Acute subdural hematoma, Acute intracerebral hematoma |
|
|
Contusion
|
Area of cortical hemorrhage at the surface of brain.
The contusion may be loacted beneath the site of impact (Coup Lesion) or distant from the site of impact (Contrecoup Lesion). Contusions may cause focal neurologic deficits & may later become seizure foci. |
|
|
Acute epidural hematoma
|
Rapidly accumulating blood clot due to skull fracture & tearing of middle meningeal artery
Causes a mass effect |
|
|
Acute subdural hematoma
|
A rapidly accumulating blood clot due to tearing of the veins "bridging" the subdural space.
Causes a mass effect |
|
|
Acute intracerebral hematoma
|
Rapily accumulating blood clot within brain parenchyma due to vascular disruption within brain.
The vascular disruption results from "shearing" forces generated at time of impact |
|
|
Hematoma Diagram
|
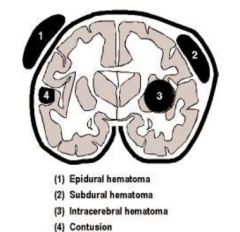
|
|
|
Diffuse Brain Injury
|
Head trauma, especially that associated with automobile accidents, may cause diffuse brain injury due to shearing forces generated by deceleration/acceleration. Diffuse brain injury results in global brain dysfunction rather than the localized neurological deficits typically associated with focal brain injuries. With diffuce brain injury, the patient is unconscious from the moment of impact.
May be Concussion or Diffuse Axonal Injury |
|
|
Concussion
|
A clinical syndrome of transient physiological dysfunction evidenced clinically by a short-term loss of consciousness
|
|
|
Diffuse Axonal Injury
|
Axonal disruption resulting from the shearing forces generated within the brain at the time of impact
|
|
|
Complications of Head Trauma
|
1. Brain edema - magnified any mass effect
2. Mass lesions - brain shifts, herniation, & death 3. Raised intracranial pressure (ICP) - leads to decreased cerebral perfusion pressure & consequent global brain ischemia 4. Ischemic brain injury - from hypotension, raised ICP, etc 5. Infection - meningitis or brain abscess 6. Post-traumatic epilepsy |
|
|
2 Types of Strokes:
|
Ischemic or hemorrhagic
1. Brain infarct - due to loss of blow flow (ischemic stroke) --> the cause of 80% of strokes 2. Intracerebral hemorrhage - due to arterial rupture (hemorrhagic stroke) --> cause of 10% of strokes 3. Subarachnoid hemorrhage - due to arterial rupture (hemorrhagic stroke) --> cause of 10% of strokes |
|
|
Stroke Diagram
|
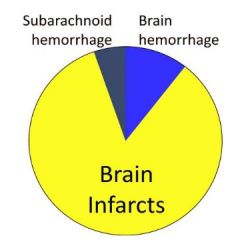
|
|
|
Hodgkin lymphoma
|
-malignant lymphoma characterized by a distinctive cell: the Reed-Sternberg cell
-origin of Reed-Sternberg cell was controversial (convincingly of B-cell origin) -EBV genome is present in up to 50% of cases -peak incidence in 20's, smaller peak at age >50 years (bimodal age distribution) -extranodal involvement is unusual -spreads along contiguous lymph node chains (unlike NHL(non-hodgkins lymphoma)) |
|
|
Pathologic diagnosis of Hodgkin Lymphoma
|
-diagnosis based on finding Reed Sternberg cells in the appropriate cellular background: lymphocytes, eosinophils, histiocytes, fibroblasts
-Morphologic subtypes correclate with prognosis -nodular sclerosis = good prognosis -lymphocyte predominance= very good -mixed cellularity -intermediate -lymphocyte depletion- worst prognosis -therapy has largely equalized these discrepancies. therefore HD has good prognosis |
|
|
Hodgkin Stage I
|
involvment of single lymph node region or single extrolymphatic site (l E)
|
|
|
Hodgkin Stage II
|
involvement of two or more lymph node regions on same side of diaphragm; may include localized extralymphatic involvemetn on same side of diaphram (II E)
|
|
|
Hodgkin Stage III
|
-invovlement of lymph node regions on both sides of the diaphragm; may include spleen (IIIs) or localized extranodal disease (III E)
|
|
|
Hodgkin Stage IV
|
-diffuse extralymphatic disease (in liver, bone marrow, lung, skin)
|
|
|
Hodgkin lymphoma clinical staging
|
5 year survival based on stage: stage is more important than histologic subtype (unlike NHL)
Stage I and II- 90% Stage III-84% Stage IV - 65% |
|
|
Hodgkin lymphoma imaging
|
-CT scan of chest, abdomen and pelvis
-PET scanning for baseline and follow-up -resistant disease detectable with PET PET currently the best predictor: Negative PET after chemo x2 95% PFS at two years Positive PET after chemo x2 10% PFS at two years |
|
|
Hodgkin Lymphoma therapy
|
easly stage issues/options
rxt/chemo vs. chemo alone ABVD (adriamycin, bleomycin, vinblastine, dacarbazine) is treatment of choice : low recurrence, few side effects |
|
|
Advanced stage Hodgkin lymphoma therapy
|
(IIB, III, IV)
-chemoterapy- ABVD -hybrid/ other regimens -consolidation with rxt If relapsed: hematopoietic stem cell transplant |
|
|
Non-hodgkin lymphomas
|
-malignant clonal diseases of lymphocytes
-NOT stem cell disorders -many subtypes: remember there are B-T and NK cells -several classification schemes -working formulation is used for clinical classification (1982) -World Health Organization (WHO) is used for pathologic classification |
|
|
Classification goals
|
-to group patients into prognostic groups
-to be easily/reproducibly used by pathologists -Historically based on histology alone: -architectural arrangement -cell size and shape |
|
|
Precursor B cell neoplasm
|
-precursor b-cell lymphoblastic leukemia/lymphoma (b-cell acute lymphoblastic leukemia) lukes collins
|
|
|
Mature (Peripheral) B cell neoplasms
|
-B cell chronic lymphocytic leukemia/small lymphocytic lymphoma
-lymphoplasmacytic lymphoma (Waldenstrom's macroglobulinemia) -hairy cell leukemia -plasma cell myeloma/plasmacytoma -extranodal marginal zone B-cell lymphoa of MALT type -follicular lymphoma -mantle cll lymphoma -diffuse large B-cell lymphoma -burkitt lymphoma |
|
|
Precursor T-cell neoplasm
|
-precurso T-cell lymphoblastic leukemia/lymphoma (t-cell ymphoblastic lymphoma)
|
|
|
Mature T-cell (peripheral) neoplasms
|
-T-cell granular lymphocytic leukemia
-adult t-cell lymphoma/leukemia (HTLV1+) -Enteropathy-type t-cell lymphoma -mycosis fungoides (sezary syndrome) |
|
|
Small lymphocytic Lymphoma
|
-essentially the same disease as CLL (chronic lymphocytic leukemia)
-SLL (small lymphocytic lymphoma) and CLL both are: -comprised of small, round lymphocytes -have the same immunophenotype -occur in older adults -are considered incurable -Mature B-cell (CD 19+, CD 20+, CD 23+) co-expressing CD5, kappa or lambda light chain |
|
|
follicular lymphoma
|
-midle-aged to elderly adults
-cells resemble normal germinal center cells -mature B-cell immunophenotype -Cytogenetics: t (14;18) -translocates bcl-2proto-oncogene downstream of immunoglobulin regulatory sequences |
|
|
Follicular lymphoma low grade histology
|
-indolent course- median survival 10 years
-incurable with conventional chemo -pts typically present with high stage disease (including marrow involvement) -may progress to difuse large cell lymphoma -mature B-cell phenotype (CD19,20 +) CD 10 + (immature B-cells and follicle center cells) kappa or lambda light chain |
|
|
diffuse large cell lymphoma
|
-most common type of NHL
-intermediate grade lymphoma -low stage disease can be cured -occurs in children and adults -can be associated with immune dysfunction (HIV, SCID, BM & organ transplants) -1/3 cases are extranodal (GI< Brain) -most often mature b-cell phenotype |
|
|
Burkitt lymphoma
|
-endemic (africa;98% EBV genome positive)
-predominantly in children -Non-endemic (worldwide: 15-20% EBV positive) all ages -Cytogenetics: t(8;14) most common but also t(2;8) & t(8;22) -c-myc proto-oncogene downstream of immunoglobulin heavy chain regulatory sequences -mature B-cell phenotype |
|
|
classification of Non-hodgkin review
|
Working formulation based on clinical grade (low, intermediate, high)
WHO based on phenotype and maturity -precursor (immature) and mature B-cell neoplasms -precursor (immature) and mature T-cell neoplasms |
|
|
Development of B-cell
|
lymphoblast --> B-lymphocyte: (naive CD19/CD10 IgM/IgD)---> Plasma cell----> CD 138
|
|
|
Plasma cell dyscrasia
|
-clonal disorders of terminally differentiated B-cells
-Clinical presentation: variable; features resultant of organ dysfunction;lytic lesions or fractures in 70% cases at diagnosis -10-15% of hematologic malignancies (20,000 cases/year in US) -mean age: 60-'s -Men> Women -significant racial bias: AA: Cauc = 2:1 |
|
|
Classification of Plasma Cell dyscrasia depends on ...
|
Identification and characterization of clonal plasma cell population
-paraprotein analysis (classically electrophoresis) -morphology -immunophenotype (IHC/flow cytometry -Cytogenetic/molecular techniques Clinical Radiographic findings -clinical presentation -sites of involvement -extent of disease |
|
|
Current diagnostic criteria for plasma cell myeloma
|
-M protein in serum or urine
-clonal plasma cells in bone marrow or plasmacytoma -Related organ or tissue impairment C: hypercalcemia R: renal insufficiency A: anemia B: bone lesions |
|
|
clonal hematologic malignancies
|
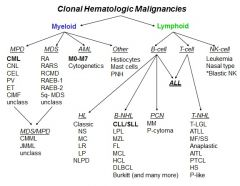
|
|
|
Lymphomas different types
|

|
|
|
Cause of Brain Ischemia
|
-Arterial occlusion due to ATHEROMATOUS PLAQUE or THROMBUS. Extracranial carotid arteries are common sites fro atheromatous plaques.
-Arterial occlusion due to an EMBOLUS. Embolus may arise from thrombotic material on an atheromatous plaque in the aorta or extracranial carotid arteries, from a mural thrombus in teh heart, from thrombotic material (vegetations) on heart valves,etc -CARDIAC ARREST or severe SYSTEMIC HYPOTENSION can cause global (diffuse) ischemic brain damage |
|
|
Consequences of Brain Ischemia
|
-NO EFFECT - anastomoses (circle of Willis, leptomeningeal vessels, etc) may provide sufficient blood flow to the ischemic area
-TRANSIENT ISCHEMIC ATTACK (TIA) - clinically evidenced as a transient neurologic abnormality. TIAs are reversible & are not accompanied by irreversible brain damage, i.e., infarcts, but may herald a future infarct -INFARCT - ischemic brain undergoes necrosis, atrophy & cavitation. The neurons (gray matter) are most vulnerable to ischemic injury. |
|
|
Macroscopic & Microscopic evolution of an infarct
|
-Coagulative necrosis (red neurons) is present at 12-24 hrs.
-Influx of neutrophils within infarcted tissue occurs by 24 hrs. -Influx of macrophages occurs by 3rd day -Edema & mass effect is maximal in 1st week & waves thereafter -Glial proliferation (gliosis, glial scarring) occurs in brain tissue at the periphery of the cavity -Cavitation of infarct takes weeks to months, depending on size of infarct |
|
|
Location of Brain Infarct
|
Often provides a clue as to its cause
Locations: Cortical infarct "Borderzone" infarct "Lacunar" infarct Global ischemic injury |
|
|
Cortical Infarct
(Location of Brain Infarct) |
Due to occlusion of a large artery.
Usually caused by atherothrombosis or embolism |
|
|
"Borderzone" Infarct
(Location of Brain Infarct) |
Occurs int he borderzone region between 2 arterial distributions.
Due to an episode of systemic hypotension |
|
|
"Lacunar" Infarct
(Location of Brain Infarct) |
Due to occlusion of a small penetrating artery.
These deep infarcts are usually cavitary (cystic) |
|
|
Global Ischemic Injury
(Location of Brain Infarct) |
Aka Global Ischemic Encephalopathy
Due to prolonged cardiac arrest Ischemic injury is widespread |
|
|
Consequences of a Brain Infarct
|
-Brain necrosis leads to NEUROLOGIC DEFICIT of rapid onset. The type of neurologic de3ficit is determined by the location of the infarct
-Possible MASS EFFECT from edema within infarcted tissue -DEMENTIA ("vascular dementia") may complicate multiple brain infarcts |
|
|
Causes of nontraumatic (spontaneous) Brain Hemorrhage
|
-HYPERTENSION - the common cause of nontraumatic brain hemorrhage
-Bleeding disorder -Rupture of an arteriovenous malformation -Drug abuse (e.g., cocaine, amphetamines) -Cerebral amyloid angiopathy - occurs in other patients -Hemorrhage into primary or metastasis neoplasm in brain |
|
|
Berry (Saccular) Aneurysms
|
Arise on large arteries in the subarachnoid space at the base of the brain (e.g., arteries of the circle of Willis)
-Aneurysms arise at the forks of arteries -Prevalence of aneurysms in the general population is 2-5% in adults, with higher frequency in patients with polycystic kidney disease or coarctation of the aorta -Berry aneurysms are frequently multiple (20%) |
|
|
Consequences of massive Subarachnoid Hemorrhage from Ruptured Aneurysm
|
-Hemorrhage causes increased intracranial pressure
-Hemorrhage may also jet upward into brain parenchyma & produce a localized hematoma with a mass effect -Ruptured aneurysm may rebleed -Subarachnoid hemorrhage may cause arterial vasospasm & infarcts |
|
|
Consequences of a Brain Tumor
(Diagram) |
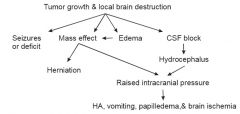
|
|
|
All Intracranial Tumors are potentially lethal
(Diagram) |

|
|
|
Histological Classification of tumors of the nervous system
(Differentiation of Tumor Cells --> Histological Classification) |
Glial differentiation --> Astrocytoma & glioblastoma, Oligodendroglioma, Ependymoma
Neuronal differentiation --> Medulloblastoma Arachnoidal-cell differentiation --> Meningioma Schwann-cell differentiation --> Schwannoma & neurofibroma, Malignant peripheral nerve sheath tumor (MPNST) Miscellaneous types of differentiation --> Pituitary adenoma; Lymphoma, primary or metastatis; Hemangioblastoma; Craniopharyngioma; Germ-cell neoplasms of pineal & suprasellar areas Metastatic tumors --> Carcinomas, sarcomas, etc. |
|
|
Incidence of Intracranial Tumors
(Diagram) |
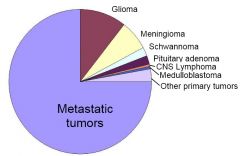
Note that gliomas, metastases & meningiomas make up the majority of brain tumors
|
|
|
Clinical & Pathologic Correlations
(Brain Tumor locations and types of neoplasms) |

|
|
|
Malignant Glioma (Glioblastoma)
|
Very poorly differentiated astrocytomas, with mitotic figures, necrosis, & a fast rate of growth.
Their infiltrative nature precludes complete surgical excision, giving them a very poor prognosis |
|
|
Meingioma
|
Meningiomas are external to the brain & do not infiltrate underlying brain
These histologically benign tumors rarely metastasize & can often be completely surgically excised, giving them a good prognosis |
|
|
Schwannoma (Vestibular schwannoma)
|
Arising on the left 8th cranial nerve.
These benign tumors are well circumscribed & can be completely excised, giving them a good prognosis The patient presents with vertigo or hearing loss, reflecting involvement of the 8th cranial nerve |
|
|
Relative Frequency of Primary Neoplasms Metastatic to the Brain
|
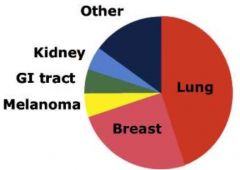
|
|
|
"Demyelinating Disease" Definition
|
1. Difference between Demyelination, where myelin breakdown occurs but the axons are preserved; & axonal (Wallerian) degeneration, where myelin breakdown occurs secondary to degeneration of the underlying axons.
2. There is potential for REMYELINATION & AXONAL REGENERATION in PNS, but no significant potential in CNS. |
|
|
Demyelination & Remyelination in PNS
(Diagram) |
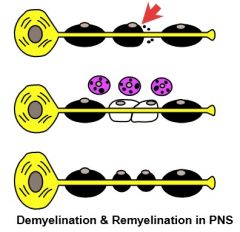
|
|
|
Wallerian Degeneration & Axonal Regeneration
|
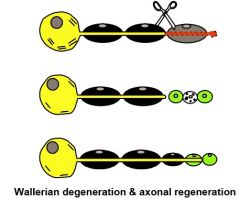
|
|
|
Mechanisms by which myelinating glial cells of the nervous system are selectively injuried (selective vulnerability)
|
1. Immune-mediated (multiple sclerosis, CNS; Guillain-Barre syndrome, PNS)
2. Viral infection of oligodendrocytes (progressive multifocal leukoencephalopathy) 3. Inherited metabolic defects (the leukodystrophies) 4. Toxins (diphtheria neuropathy) |
|
|
Multiple Sclerosis (MS)
|
An immune-mediated demyelinating disease of CNS
1. Occurs between 15-50 years of age, with average age of onset at 30 2. Currently considered an autoimmune disease in which environmental factors & heredity contribute to risk 3. Pathology is that of demyelination plus the chronic inflammation typical of an autoimmune disease |
|
|
Progressive Multifocal Leukoencephalopathy (PML)
|
Another type of CNS demyelinating disease
The viral disease, which typically occurs in immunocompromised patients, is most commonly found in AIDS patients. Unlike MS, the demyelination is accompanied by viral inclusions in oligodendrocytes & bizarre reactive astrocytes. |
|
|
Leukodystrophies
|
Inherited metabolic diseases in which the myelinating glia (oligodendrocytes & sometimes Schwann cells) are severely affected because of an enzyme defect, which prevents the normal catabolism of myelin.
|
|
|
Alzheimer's Disease
|
-Responsible for 50-60% of the cases of dementia
-Characterized primarily by a degeneration of the neurons in the CEREBRAL CORTEX & HIPPOCAMPUS, which causes dementia -Etiology & Pathogenesis = Unknown. Most adults with Down syndrome develop the pathological changes of Alzheimer's disease by age 40. Most cases are sporadic, but a small percent of cases are familial & the genes have been identified (beta amyloid precursor protein gene; presenilin 1 & 2 genes). Apolipoprotein E4 is an established risk factor. |
|
|
Alzheimer's Disease
(Degenerative Changes in the Cytoskeleton of involved neurons, in the neuropil of the gray matter & in cerebral blood vessels) |
-NEUROFIBRILLARY TANGLES in neurons --> composed of intracytoplasmic paired helical filaments (PHF) in the neuronal cytoplasm. PHF are an abnormally phosphorylated isoform of TAU, a low molecular weight microtubule associated protein
-SENILE PLAQUES in the neuropil - composed of extracellular amyloid (amyloid Beta protein) surrounded by PHF-filled neuronal processes. -Vascular deposits of amyloid Bta protein (Cerebral Amyloid Angiopathy) |
|
|
Huntington's Disease
|
-Autosomal Dominant Inheritance. The mutated gene is on chromosome 4 & encodes for the protein, huntingtin. The mutation is an expanded CAG repeat. Clinical onset usually around 20-50 years of age. Characterized by progressive choreiform movements & dementia
-The neuronal degeneration is primarily in the basal ganglia (which produces chorea) & in the cerebral cortex (which produces dementia) -Histopathologically, there are loss of the smaller neurons in the caudate nucleus & putamen, & loss of neurons in the cerebral cortex |
|
|
Parkinson's Disease
(Characterizations & Pathogenesis) |
-A neurogenerative disease characterized mainly by a degeneration & loss of the pigmented neurons of the substantia nigra in the midbrain. The loss of pigmented cells is so great that it can be seen grossly. The loss of neurons in the substantia nigra causes the clinical picture of parkinsonism
-The pathogenesis of this sporadically occuring disease is unknown. Rare familial forms do occur & have been linked to several different gene mutations |
|
|
Parkinson's Disease
(Histopathology & Other Diseases) |
-The degenerating neurons show characteristic LEWY BODIES, which are composed primarily of alpha-synuclein. Lewy bodies are characteristic of Parkinson's disease, but can be seen in patients without Parkinson's disease
-Other disease processes can also produce tremor, rigidity, & bradykinesia, and thus a Parkinson-like picture clinically. Among the diseases associated with the parkinsonism syndrome are certain drugs (phenothiazines, etc), postencephalitic parkinsonism, infarcts of the substantia nigra, & certain other neurodegenerative diseases. |
|
|
Infectious Diseases of the Nervous System
|
A wide variety of infectious agents may invade the nervous system. Some have a predilection for the CSF & cause a leptomeningitis, whereas others preferentially infect various parts of the brain (encephalitis) or spinal cord (myelitis). Infectious agents may get to the brain via the blood stream, by direct spread from an adjacent focus of infection in the scalp, skull, or paranasal sinuses, or by direct implantation during trauma. Opportunistic agents are becoming increasingly important causes of CNS infection because of the increasing prevalence of individuals with compromised immune systems. Infections cause CNS dysfunction by destruction of brain tissue as well as by causing a mass effect.
|
|
|
Acute Bacterial Meningitis (Leptomeningitis)
|
-An acute inflammation of the leptomeninges (pia & arachnoid)
-Pathogenesis of the CSF infection (Bacteremia from a known or unknow distant focus of infection; Direct extension of infection from middle ear, mastoid bone, paranasal sinuses, or head wound) -Bacteriology of acute bacterial meningitis (Neonate - Group B Strep, Listeria, enteric gram-negative rods; Child - S. pneumoniae, N. meningitidis, H. influenzae; Adult - S. pneumoniae, N. meningitidis) |
|
|
Clinico-pathologic Correlations of Acute Bacterial Meningitis
|
-Meningeal inflammation --> stiff neck, headache
-"Toxins" --> brain swelling --> increased ICP --> herniation -"Toxins" --> encephalopathy & seizures -Inflammation of cranial nerves --> nerve palsies -Thrombophlebitis --> brain infarcts -Meningeal fibrosis --> hydrocephalus |
|
|
Chronic Meningitis
|
May complicate infections with a protracted clinical course, such as tuberculosis, certain fungal infections & syphilis.
When these chronic infections involve the leptomeninges, a chronic inflammatory process ensues. This chronic inflammatory process in the subarachnoid space also affects the cranial nerves & blood vessels in the subarachnoid space, leading to cranial nerve palsies & brain infarcts |
|
|
Brain Abscess
|
A pus-filled cavity walled off by granulation tissue
|
|
|
Pathogenesis of Brain Abscess
|
-Bacteremia from distant focus of infection
-Traumatic implantation -Direct extension from infection in middle ear, sinuses, etc |
|
|
Bacteriology of Brain Abscess
|
Often a mix of aerobic & anaerobic bacteria
|
|
|
Clinico-Pathologic correlations of a Brain Abscess
|
-Local brain destruction --> neurologic deficit
-Mass effect --> herniation --> death -Extension into ventricle --> ventriculitis & death |
|
|
Viral Infections of the CNS
(Pathogenesis of Viral Meningitis or Viral Infection) |
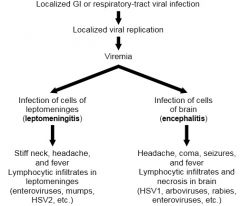
|
|
|
HSV 1 Encephalitis
|
-The most commonly occurring sporadic viral encephalitis.
-Viral encephalitis is very destructive, causing necrosis & hemorrhage in the brain. -Microscopically, the necrosis is accompanied by lymphocytic inflammation and, with some viruses, characteristic intranuclear or intracytoplasmic viral inclusions. |
|
|
Fungal Infections of the CNS
|
These "opportunistic" infections usually occur in immunocompromised patients & may cause a chronic meningitis (e.g., Cryptococcus neoformans) or encephalitis (e.g., Candida species)
|
|
|
Spongiform Encephalopathies (Prion Diseases)
|
Creutzfeldt-Jacob disease
|
|
|
Neuropathology of AIDS
|
-HIV Encephalitis (subacute giant-cell encephalitis)
-Opportunistic infections - such as CMV encephalitis, PML, cerebral toxoplasmosis, & cryptococcal meningitis -CNS lymphoma -AIDS-related myelopathy & peripheral neuropathy |
|
|
Diagnosis and classifications (and ultimately management) of hemtopoietic neoplasms is dependent upon
|
-clinical manifestations,
-morphology of biopsied tissue (special stains) -flow cyometry -molecular studies - cytogenetics -findings guide therapy, serve as upfront prognostic markers, and predict response to treatment -each technology has its strengths and weaknesses |
|
|
Development and Differentiation
|
-patterns of gene expression vary as cells mature and differentiate
-morphological differentiation -differences in expression can be exploited to help classify disease |
|
|
Cluster of Differentiation: The CD designation
|
-fisrt est. in 1982
-currently ~350 designated surgace antigens -Commonly encountered markers: CD45 |
|
|
CD45
|
-all normal leukocytes to vary degrees
-LCA=leukocyte common antigetn |
|
|
B Cell CD designation
|
- CD10+
-CD19+ -CD20+ -CD22+ -CD79a K/LPAX-5+ |
|
|
T cell CD designation
|
-CD2+
-CD3+ -CD4+ -CD5+ -CD7+ -CD8+ |
|
|
NK cell CD designation
|
-CD16+
-CD56+ |
|
|
Plasma Cell CD designation
|
-CD38+
-CD138+kappa/lambda |
|
|
Monocytes CD designation
|
-CD4+
-CD64+ |
|
|
"Blasts" CD designation
|
-CD34+
-CD117+ |
|
|
Megas CD designation
|
-CD41+
-CD42+ -CD61+ -Factor VIII+ |
|
|
Myeloid CD designation
|
-MPO+
-CD10+(mature) -CD15+ -CD33+ |
|
|
Immunophenotyping
|
-Low numbers: T-cell phenotype:remember CD4 counts and HIV/AIDS-low T cells (CD 1,2,3,4,5,7,8)
-Mid teens=Myelomonocytic (CD13=granulocyte, CD14=monocytic, CD15=granulocyte) -Nineteen and early twenties: B-cell phenotype(CD19,20,21,22,23) -Other markers (CD34 stem cell, CD33 myeloid) |
|
|
Overview of Flow Cytometry
|
-measurement of cellular properties as they are moving in a fluid stream, past a stationary set of detectors
-Detectors: Positional and Fluorescence -Positional (forward scatter {size}, side scatter {complexity}) -Fluorescence; attach a fluorescent probe to an antibody directed at a specific marker, excite with a light, measure emission, immunophenotype -FACS - fluorescence activated cell sorting |
|
|
Immunophenotyping
|
-identifying the phenotype using fluorescently labeled antibodies as probes
|
|
|
Neoplastic Hematopathology
|
-leukemia (acute and chronic)
-Lyphoma (hodgkin and non-hodgkin) |
|
|
Classification of Leukemias
|
-diagnosed according to prominent cell type involved
-Acute: rapid onset, aggressive, usually poorly differentiated (blasts) -Chronic: insidious onset, usually less aggressive and more mature appearing |
|
|
Acute Lymphoblastic Leukemias
|
-lymphoid precursor (lymphoblast
|
|
|
Acute Myeloid Leukemias
|
-myeloid precursor (myeloblast
|
|
|
Chronic Lymphoproliferative Disorders
|
-Lymphocyte (B, T, NK)
-Plasma cell |
|
|
Myeloproliferative Disorders and Myelodysplastic Syndromes
|
-Granulocytes
-basophils -eosinophils -platelets -monocytes |
|
|
Chronic Myeloid Leukemia (CML)
|
-myeloproliferative neoplasm that originates in bone marrow stem cell associated with BCR-ABL1 fusion gene
-20-40% of pts are symptomatic and diagnosed incidentally -WBC: leukocytosis with blood that looks like a bone marrow aspirate -numerous immature granulocytes -increased basophils -hypercellular -increased M:E |
|
|
Benign neutrophilia
|
-many etiologies left shift not marked as in CML (may see bands but infrequently see myelocytes and other immature forms)
|
|
|
BCR-ABL fusion Gene
|
-juxtapostions of BCR (22) with ABL(9)
-constituatively avtive tyrosine kinase (upregulating cell cycling) -karotype, FISH, PCR -Target for therapy -normal karotype doesnt exclude a cryptic translocation |
|
|
Leukemia progression
|
-untreated, the chronic phase disease will ultimately transform to a blast phase with :
*additional genetic abnormalities *increased blasts -Variants: AML and ALL (remember where the defect is) |
|
|
Chronic Lymphocytic Leukemia (CLL)
|
-kidn of an outlier from the other leukemias
-disease equivalent to small lymphocytic lymphoma (SLL, tissue vs bone marrow/blood based) -mature B cell lymphoma -pts often older and die with disease and not from it -more agressive subsets exists -50% have hypoammaglobulinemia (infection) Coombs + AIHA, ITP -can have involvement of the bone marrow, lymph nodes (SLL), spleen(white pulp), etc |
|
|
Leukemia Immunophenotype
|
-Typical pattern:Mature B cell phenotype expressing and CD5 and CD23 (CD19+CD20+(weak)CD5+CD23+)
-Weak sIgM or IgM/IgD+ -light chain restricted -CD10-Cylcin D1-CD103 -CD10: follicular, burkitt, ALL -Cyclin D1: mantle cell lymphoma (CD5+ but CD23-) -CD103: Hairy Cell leukemia |
|
|
Cytogenetics
|
-Deletion 13q14 (found in >50% cases, good prognosis, better than nl karyotype, microRNA and BCL-2)
-Trisomy 12 (found in ~20% cases,worse prognosis, atypical morphology and IPT {FMC7+, CD38+}, unmutated IgVH) -Deletion 11q23(~20%cases, bad prognosis, ATM, MLL and others) -Deletion 17p13 (5% of untreated cases, bad prognosis, less responsive to therapy, p53) -Deletions of 6 (Increased PL, bad prognosis) |
|
|
Acute Myeloid leukemia
|
-Clonal expansion of meyloid blasts
-classification requires (>/=20% of blood or marrow blasts, Auer rods are seen only in a subset of cases) -Can arise de novo or as a consequence of underlying disorder such as MDS, MPD |
|
|
Acute Myeloid WHO classification
|
-Acute Myeloid with recurrent genetic abnormalities (15/17; PML/RAR alpha; Acute promyelocytic leukemia)
- Acute myeloid not otherwise specified : rest of FAB classification -Acute myeloid with multilineage dyplasia:following MDS or MDS/MPD or w/o antecedent MDS -Acute meyloid and myelodysplastic, therapy related (alkylating agent-related, topoisomerase type II inhibitor related) |
|
|
t(15;17)(q22;q12); PML/RAR alpha Acute Promyelocytic Leukemia
|
-blocked at promyelocytic stage of granulocyte differentiation
0comprised 5-8% of AML -Clinically associated with DIC -95% with reciprocal translocation t(15;17), with fusion of PML gene on 15 and retinoic acid receptor alpha (RAR alpha) on 17 -Remaining 5% show RAR alpha rearrangements with other genes |
|
|
t(15;17) and ATRA
|
-Promyelocytic leukemia (PML) protein :tumor suppressor that forms complexes with other proteins (P53, pRb, and others)
-Retinoic acid receptor alpha -all-trans retinoic acid (ATRA): RA analogue:overcomes the avidity of RAR alpha binding to corepressor complex, differentiation therapy |
|
|
Retinoic Acid Receptor Alpha
|
-Normally heterodimerizes with retinoid X receptor (RXR) and interacts with nuclear corepressor complex to transcriptionally block granulocyte differentiation
-Retinoic acid release teh complex and allows progression of differentiation past the promyelocyte stage |
|
|
AML with Normal cytogenetics
|
-Approx. 50% of AML's have no clonal cytogenic abnormality
-clearly not devoid of genetic mutations -Mutations at genetic level: (FLT3, NPM1, RAS, CEBPA, etc) |
|
|
AML w/normal cytogenetics: FLT3
|
-FMS-like tyrosine kinase 3
-mutations occur in 20-30% of de novo AML -most frequent molecular abnormality in AML -Associated with POOR prognosis -internal tandem duplication=most common form |
|
|
Acute Lymphoblastic Leukemia (ALL)
|
-variable clinical presentation
-most common in kids -frequently have peripheral blood leukocytosis with numberous circulating blast (but can be aleukemic) -most commonly of B-cell origin, variable prognosis but generally yes, much better in children than adults -less commonlyTcell (T-ALL) mediasinal or soft tissue masses,consideed higher risk, prognosis worse tahat B-ALL |
|
|
ALL Cytogenetics
|
-karyotype and specifically targeted fluorescence in situ hybridization (FISH) and molecular studies
-B-cell acute lymphoblastic leukemia( cytogenetic abnormalities seen in majority of cases, some are recurrent, some have significant prognostic implications) -T-cell acute lymphoblastic leukemia:clonal rearrangement of T-cell receptor gene |
|
|
Favorable prognosit cytogenetic classifications of adult ALL
|
-Favorable: hyperdiploid and t(12;21)
-Unfavorable:t(9;22), abn (11q23), hypodiploid |
|
|
ALL
|
-common in kids, good prognosis
-less common in adults, worse prognosis -lymphoblasts large cells with open chromatin -t(9;22) bad but doesnt independently drive diseae |
|
|
AML
|
-look for incerased blasts and Auer rods
-cytogenetics subtype increasingly more important |
|
|
CML
|
-t(9;22) BCR-ABL drives disease (not the most accurate representation of malignancy)
-Can transform to acute leukemia (lymphoid/myeloid) |
|
|
CLL
|
-older pts
-CD20+CD19+CD5+CD23+flow is a great starer tooth -differentiate from mantle and cell lymphoma |
|
|
Etiology
|
Cause of a disease process
|
|
|
Pathogenesis
|
Mechanisms of disease development
|
|
|
Morphologic changes
|
Structural changes due to disease
|
|
|
Clinical findings
|
Functional consequences of disease
|
|
|
GI Tissue Layers
(Diagram) |
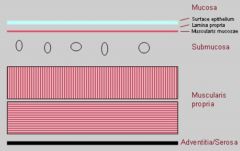
|
|
|
Esophagitis
|
Defined: Inflammation of esophageal mucosa
Prevalence: 5% of US Types: -Reflux esophagitis (aka GERD) -Infectious esophagitis -Barrett esophagus -Other: radiation, chemical |
|
|
Reflux Esophagitis
(Etiology & Pathogenesis) |
Etiology --> Gastric contents reflux back into esophagus
-Decreased lower esophageal sphincter (LES) tone -Hiatal hernia -Increased intra-abdominal pressure Pathogenesis --> Gastric fluid causes esophageal mucosal damage |
|
|
Reflux Esophagitis
(Decrease in tone of lower esophageal spincter - LES) |
-Fatty meals
-Alcohol -Smoking -Presence of nasogastric tube -Pregnancy (progesterone) |
|
|
Reflux Esophagitis
(Increase in luminal pressure) |
-Large meals
-Obesity -Pregnancy (mass effect) -Bending at the waist |
|
|
Reflux Esophagitis
(Morphologic changes) |
-Inflammatory cells in esophageal squamous epithelial (Lymphocytes, eosinophils, neutrophils)
-Basal zone hyperplasia -Elongation of lamina propria papilla -Spongiosis |
|
|
Reflux Esophagitis
(Clinical Findings) |
-Dysphagia
-Heartburn/chest pain -Hematemesis |
|
|
Reflux Esophagitis
(Consequences/complications) |
-Bleeding
-Ulceration -Stricture (Deep ulcers can heal with scarring leading to narrowing of the esophageal lumen) -Barrett esophagus |
|
|
Infectious Esophagitis
(Etiology & Pathogenesis) |
Etiology:
-Fungal - candidiasis -Viral - Herpes & CMV Pathogenesis: -Inflammation & resulting tissue damage in response to infectious viral or fungal agent |
|
|
Infectious Esophagitis
(Morphologic changes) |
Candida:
-Gross - white/gray adherent pseudomembranes -Micro - yeast & pseudohyphae Herpes virus: -Gross - punched-out ulcers -Micro - HSV & CMV have characteristic viral cytopathic effect |
|
|
Herpetic Viral Cytopathic Effect
|
-Multinucleated squamous epithelial cells
-Margination of normal chromatin forming a peripheral rim with central ground glass appearance -Nuclear Moulding |
|
|
Infectious Esophagitis
(Clinical findings) |
-Immunosuppressed patients
-Dysphagia -Pain |
|
|
Barrett Esophagus
(Definition) |
Replacement of the distal esophageal squamous epithelium by metaplastic columnar epithelium (with intestinal type goblet cells)
|
|
|
Barrett Esophagus
(Etiology & Pathogenesis) |
Etiology --> Chronic reflux injury (GERD)
Pathogenesis --> Injury from reflux of gastric acid results in metaplasia: -Conversion of one differentiated cell type to another -Benign, adaptive process |
|
|
Barrett Esophagus
(Morphologic Changes) |
Replacement of the squamous epithelium of the distal esophagus with columnar epithelium containing goblet cells
|
|
|
Barrett Esophagus
(Diagnosis: 2 Criteria) |
1. Endoscopic findings (red, velvety mucosa)
-Histologic findings of columnar mucosa with goblet cells in biopsy material from specified location ("intestinal metaplasia") |
|
|
Barrett Esophagus
(Clinical findings) |
Risk factors:
-Age >40 -Caucasian -Male -Any underlying cause for long standing reflux esophagitis Same symptoms as GERD |
|
|
Barrett Esophagus
(Complications) |
Dysplasia --> disordered cell growth
10% lifetime risk for development of Adenocarcinoma: -Risk of developing adenocarcinoma is related to the length of the metaplastic segment (30-40 fold increase risk over general population if >3cm of Barrett is present) |
|
|
Esophageal Carcinoma
|
-Uncommon (6% of all cancers in the GI tract)
-Usually asymptomatic until they cause obstruction (often too late to permit cure; without treatment, average survival is 4 months after diagnosis) -Squamous cell carcinoma is more common worldwide -Adenocarcinoma & squamous cell carcinoma exhibit comparable incidence rates in the US |
|
|
Esophageal Adenocarcinoma
(Definition, Etiology, & Pathogenesis) |
Definition --> Malignant epithelial tumor with gland formation
Etiology --> Majority arise in Barret esophagus; ???Helicobacter pylori??? Pathogenesis --> Multistep molecular "hits" |
|
|
Transition from Barrett Esophagus to Adenocarcinoma
|
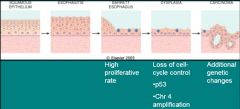
|
|
|
Esophageal Adenocarcinoma
(Morphologic changes) |
-Gross: Flat or raised lesion
-Micro: Infiltrating glands with mucin production |
|
|
Esophageal Adenocarcinoma
(Clinical findings) |
Usually asymptomatic until luminal obstruction
-Difficulty swallowing -Weight loss Diagnosis often too late to permit cure -<20% 5 year survival |
|
|
Esophageal Squamous Cell Carcinoma
(Definition, Etiology, & Pathogenesis) |
Definition --> malignant epithelial tumor with squamous differentiation
Etiology --> Diet (Vitamin deficiency, high nitrates, fungal contamination) & Lifestyle (ETOH & smoking) Pathogenesis: -Carcinogens (fungus & nitrosamines) -Potentiators of carcinogens (nutritional deficiency) -Stepwise accumulation of genetic alterations |
|
|
Squamous Cell Carcinoma
(Morphologic Findings) |
Gross:
-Polyploid, ulcerating, or flat tumor -Middle third Micro: -Invasive squamous epithelial nests within fibrotic stroma -Rich lymphatic system allows extensive tumor spread |
|
|
Gastric Diseases
|
Gastritis:
-Acute -Chronic (H. pylori) Peptic ulcer Tumors (Adenocarcinoma) |
|
|
Gastritis
(Definition & Etiology) |
Definition --> inflammation of the gastric mucosa
Etiology --> Numerous: -NSAID/ASA use -ETOH & smoking -Infection (Helicobacter pylori) -Autoimmune (chronic) |
|
|
Gastritis
(Pathogenesis) |
-Mucosal damage
-Increased H+ secretion with back diffusion -Decreased bicarb buffer -Reduced blood flow -Alteration to adherent mucus layer -Direct epithelial damage |
|
|
Gastritis
(Morphologic findings) |
Gross --> Acute & Chronic: Variable
-Congestion, ulceration, atrophy Micro: Acute/active --> neutrophils in lamina propria & in epithelium Chronic: -Lympocytes & plasma cells in lamina propria & in epithelium - +/- mucosal atrophy with intestinal metaplasia |
|
|
H. Pylori Chronic Gastritis
|
Most common type of chronic gastritis in US (90% of patients with chronic gastritis have H. pylori present)
Major etiologic factor in peptic ulcers |
|
|
H. Pylori Chronic Gastritis
(Pathogenesis) |
Virulence factors:
-Motility (flagella) -Urease (Releases ammonia - causes cell injury & buffers gastric acid) -Protease (Degrades protective mucus barrier) -Exotoxin (Damages epithelial cells) |
|
|
H. Pylori Chronic Gastritis
(Morphologic findings) |
Early - acute/active gastritis
-Neutrophils & microorganisms Later - chronic -Lymphocytes & microorganisms H. pylori in mucus layer |
|
|
H. pylori Infection & Disease
(Diagram) |
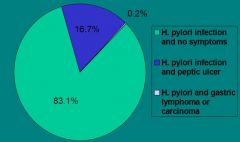
|
|
|
Laboratory Medicine
|
-clinical pathology
-the area of pathology dedicated to the selection, provision an dinterpretation of diagnostic testing for patient care -clinical chemistry, molecular diagnostics, microbiology, hematology, coagulation, transfusion medicine, cytogentics, hematopathology |
|
|
Use of Laboratory data
|
-part of medical decision process
-ordered to screen for disease, answer a question, verify disease, determine prognosis, stage, monitor an intervention or treatment -70% of all diagnosis are based upon a laboratory test result |
|
|
Screening
|
-amino acids: newborns
-PSA: prostate Cancer -cortisol: Cushings |
|
|
Diagnosis (confirmation)
|
-glucose: diabetes
-blood/urine cultures: infections -TSH: thyroid disease -hemoglobing: anemia |
|
|
Disease severity/staging
|
-PSA: prostate Cancer
|
|
|
Monitor
|
-prothrombin time: coagulation tx
-HbA1c: diabetic glycemic control |
|
|
Prognosis/risk stratification
|
-microalbumin:diabetic nephropathy
-hsCRP: cardiac |
|
|
Define "normal" reference ranges
|
-define the reference population
-without disease -exclusion criteria: recent illness, drug use (OTC, prescription, abused), obesity, etc -partioning criteria: gender, age, ethnicity, posture, fasting, time etc -95-97% of reference pop |
|
|
"normal" variatioN"
|
-ultradian (<1d)
-circadian (over 24hr) -cicannual (over year) -intra-individual variation (between days): -<5%- electrolytes, creatinine, T4 -5-10%- alkaline phosphatase, cholesterol, glucose ->10%- cortisol, ACTH, creatinine kinase, TSH |
|
|
Cholesterol
|
-I lowered my cholesterol by 9 points (200-191 mg/dL)
-4.5% analytical variability 9mg/dL -6.5% biological variability 13 mg/dL (intra-individual) |
|
|
"unexpected results"
|
pre-analytical:
-patient preparation -specimen collection an dhandling analytical variability -impression, error biological variability -physiology and unexpected pathology pharmacological effects/interactions |
|
|
Adrenal Excess
|
glucocorticoids: cushings syndrome
mineralcorticoids: Conn's syndrome Androgens: congenital adrenal hyperplasia |
|
|
Cushing's Syndrome
|
Any cause of increased glucocorticoids (cortisol)
-cushing's disease: pituitary adenoma -adrenal tumor -ectopic ACTH |
|
|
clinical features of Cushing's
|
-Cushinoid appearance: moon face, hirsutism, acne, plethora, truncal obesity, buffalo hump, edema, excessive bruising, thin skin w/ striae
-poor wound healing -hyperglycemia -osteoporosis -muscle weakness, emotional instability, cardiac hypertrophy, hypertension, polyuria |
|
|
Remember: If your patient is on a corticosteroid
|
-wounds heal poorly or slowly
-consult patients MD: may ned to increase dose, may need to delay procedures |
|
|
Thyroid
|
-two connected lobes
-12-20g (size), soft, highly vascular -synthesizes, stores and releases T4 and T3 in response to TSH -regulate and maintain overall body metabolism -promote growth and maturation -fetal brain development -organ specific actions: dec levels lead to mucoprotein deposition in skin, muscles and heart |
|
|
Regulation of thyroid hormone synthesis
|
-hypothalamus: TRH
-pituitary: TSH (establishes set point) -thyroid: T3 and T4 -periphery: 5'-deiodinase type I and type II (conversion of T4 to T3) -free T4 (FT4) about .03% of total T4 -Free T3 (FT3) about .3% of total T3 |
|
|
TSH
|
-serum TSH levels reflect the integrative action of both T3 and T4 on pituitary
-change in free thyroid hormone causes a logarithmic change in TSH |
|
|
Assessment of thyroid status
|
Purpose:
-screening: high risk groups -all newborns, women >45 y, pregnancy, history, elderly -ambulatory, "health" -doesn't apply when testing during illness -diagnosis -monitoring Key Tests -TSH (reflex to free T4, add T3 or free T3) |
|
|
Diabetes in the US
|
-21 million individuals are known to have diabetes
->1 million have Type 1 with 35-50,000 new cases/ year ->19 million have Type 2 with 6 million undiagnosed -3-5% of all pregnancies have gestational diabetes -42 million are thought to be "pre-diabetic" (have glucose intolerance) |
|
|
Type I diabetes
|
-5-10% of cases
-not age specific (onset most often in childhood or adolescence) -destruction of pancreatic beta cells -autoimmune process -causing severe insulin deficiency -80-90% destruction before symptomatic -requires physiologic replacement of insulin |
|
|
Clinical presentation Type I diabetes
|
-abrupt onset of sumptoms
-polyuria, polydipsia, rapid weight loss, ketoacidosis common -poor healing, nephropathy, neuropathies, retinopathies, hypo and hyperglycemic events |
|
|
Type 2 diabetes
|
-90% of cases (30% undiagnosed)
-complications begin before diagnosis -age, BMI, family hx, gestational diabetes, impaired glucose tolerance, phsical inactivity, race/ethnicity (all RISK factors) -insulin resistance -insulin secretory defect (may require insulin) -normal or increased insulin -poor healing, nephropathy, neuropathies, hyperglycemia |
|
|
Complications- Type I or Type 2
|
-decreased resistance to infections
-xerostomia -poor or delayed wound healing -increased incidence of periodontal disease with periodontal abscesses -before major procedures consult with patient's MD -Type 1 patients may need increased insulin -Type 2 patients may require insulin prior to treatment -both may need antibiotic therapy -during long procedures- may experience hypoglycemia -may be useful to have patient monitor glucose |
|
|
Laboratory testing
|
-glucose (plasma/serum)
-goal is to tighten glucose control but minimize hypoglycemic events -self-monitoring blood glucose -HbA1c: indicator of glucose control over last 8-12 weeks -microalbumin: used to judge renal impairment, correlates with risk of retinopathy |
|
|
Parathyroid and PTH
|
-parathyroids
-2-7mm, 50% fat -chief cells synthesize, store, and secrete PTH PTH: -direct action on bone (osteoblasts, osteocytes, and indirectly on osteoclasts) -direct action on kidney -indirect action on intestine -chief cells (small round cells with finely granular cytoplasm) |
|
|
Vitamin D
|
-dietary sources (ergocalciferols)
-vitamin D2, fish liver oils, egg yolks, liver -dermal (cholecalciferols) : 7-dehydrocholesterol ---> sunlight--> vitamin D3 |
|
|
Vitamin D synthesis
|
- (in liver) 24-hydroxylase---> 35 (OH) D: use for vitamin D status
- (in kidney) 1-hydroxylase ---> 1,25 (OH)2D: use for mineral disorders |
|
|
Osteoporosis
|
Diagnosis:
-low bone density ,fracture, physical findings (dorsal kyposis) Symptoms: -none to few (back pain from fractures) -increased caries, tooth loss |
|
|
Bisphosphonate
|
-used to treat osteoporosis
-alendronate |
|
|
Abbreviations
|
-CRH: corticotropin releasing hormone
-GnRH- gonadotropin releasing hormone -GHRH- growth hormone releasing hormone -PIF- prolactin inhibiting factor -TRH- thyrotropin-releasing hormone -GHIH- somatotropin (growth hormone) release inhibiting factor -IGF-1- insulin-like growth factor -HCG- human chorionic gonadotrophin -BMI-body mass index -DXA-dual energy x-ray absorptiometry |
|
|
Peptic Ulcer Disease
(Definition) |
Ulcer --> disruption of the mucosa that extends through the muscularis mucosa
Peptic ulcer --> distruption of the mucosa that extends through the muscularis mucosa in region of GI tract exposed to acid secretions (stomach & duodenum) |
|
|
Peptic Ulcer Disease
(Etiology) |
-Decreased mucosal defenses
-Increased mucosal damage |
|
|
Peptic Ulcer Disease
(Morphologic changes) |
Gross:
-Anterior wall first part of duodenum -Lesser curvature of stomach -Sharply punched-out mucosal defect Micro --> 4 characteristic zones: 1. Fibrinopurulent exudate 2. Necrotic tissue 3. Granulation tissue 4. Fibrotic tissue |
|
|
Peptic Ulcer Disease
(Clinical findings) |
-Epigastric pain
-Iron deficiency anemia -Hemorrhage -Perforation |
|
|
Gastric Adenocarcinoma
(Etiology/Risk Factors) |
-Dietary (Benzopyrene & nitroamines --> smoked, fried, grilled foods)
-Low SES -Helicobacter infection (antral/body tumors) -Smoking -Other --> atrophic gastritis, pernicious anemia, subtotal gastrectomy, gastric adenomatous polyps |
|
|
Gastric Adenocarcinoma
(Pathogenesis) |
Multiple genetic alterations:
-p53 mutations -E-cadherin expression alterations -TGFBRII -BAX -IGRFII |
|
|
Peptic Ulcer vs Adenocarcinoma
|
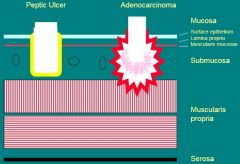
|
|
|
Gastric Adenocarcinoma
(Morphologic findings) |
2 Subtypes:
1. Intestinal --> infiltrating glands with mucin production 2. Diffuse --> infiltrating poorly differentiated single cells (signet ring cells) |
|
|
Gastric Adenocarcinoma Diffuse Type
|
Signet ring cell pattern of adenocarcinoma with cells filled with mucin & vacuoles that push the nucleus to one side
|
|
|
Celiac Disease
(Definition) |
Immune mediated disease characterized by:
-Malabsorption -Small intestine mucosal flattening -Symptomatic response to removal of gluten from diet [Gluten is a protein component (gliadin) found in wheat, oat, barley & rye] |
|
|
Celiac Disease
(Etiology & Pathogenesis) |
Etiology --> Gluten sensitivity with tissue damage
Pathogenesis: -Enterocyte damage -Failure of crypt zone cells to regenerate |
|
|
Celiac Disease
(Gross pathology & Microscopic findings) |
Gross pathology:
-Flat mucosa with loss of ridges & convolutions Microscopic findings: -Shortening in villous height -Elongation of crypts -Increased lymphoplasmacytic infiltrate in lamina propria -Histologic changes are patchy (Biopsy of multiple sites increases likelihood of finding characteristic changes) |
|
|
Celiac Disease
(Mucosal Architecture Diagram) |

|
|
|
Celiac Disease
(Surface Epithelium) |
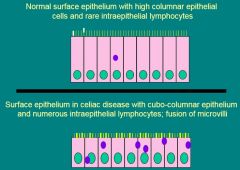
|
|
|
Celiac Disease
(Clinical Findings) |
-Usually presents in childhood
-Disease more severe in distal duodenum & upper jejunum (Decreases distally & may be minimal in distal ileum) -Severity of malabsorption dependent on length of small bowel involvement & correlates with variability in clinical presentation |
|
|
Acute Appendicitis
(Definition & Pathogenesis) |
Definition --> Inflammatory disease of the wall of the appendix
Pathogenesis: -Obstruction of the appendiceal orifice (fecalith, parasites) -Distention of the lumen & bacterial invasion |
|
|
Crypt Architecture
|
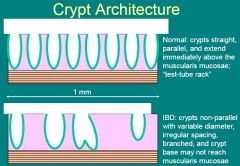
|
|
|
Inflammatory Bowel Disease
|
Idiopathic (etiology not understood)
Divided into 2 Types: 1. Crohn disease 2. Ulcerative colitis Diagnosis requires: -Chronicity -Exclusion of other etiologies (particularly infectious) |
|
|
Head Trauma
(Summary) |
Head trauma may lead to wounds of the scalp, skull fractures, large intracranial hematomas, & focal or diffuse brain lesions. Focal brain lesions are contusions, which may be "coupe" or "contrecoup," and represent localized areas of hemorrhage within the cortex. Diffuse brain injury is caused by shearing of axons within the white matter, and while not visible grossly, is a major cause of severe & irreversible brain damage. The terms "contusion" and "concussion" are often confused, but should be sharply differentiated. Traumatically induced intracranial hematomas, whether intracerebral, subdural, or epidural, are classic mass lesions & can cause devastating effects because of brain shifts & herniations. Chronic subdural hematomas often occur without clear evidence of previous head trauma & should be distinguished from acute subdural hematomas, which are typically found in patients within hours after obvious head trauma.
|
|
|
Cerebrovascular Disease
(Summary) |
Stroke = leading cause of morbidity & 3rd leading cause of mortality in US. A "stroke" may be due to an infarct in the brain, a hemorrhage within the brain, or a hemorrhage within the subarachnoid apce. Infarcts are usually due to atherosclerosis-associated intravascular thrombosis or embolism. Intracerebral hemorrhage is usually associated with hypertension or a bleeding diathesis. Rupture of a saccular aneurysm on 1 of the large arteries in the subarachnoid space at the base of the brain is the usual cause of a stroke due to a major subarachnoid hemorrhage. Both infarcts & hemorrhages lead to varying amounts of brain destruction & also act as mass lesions. The location & size of the lesion is important in determining the type & extent of the neurologic deficits precipitated by the infarct or hemorrhage.
|
|
|
Brain Tumors
(Summary) |
Brain tumors may be primary or metastatic. Gliomas are the commonest type of primary brain tumor. Menfingiomas & either-nerve schwannomas are other common primary brain tumors. Because of their location & infiltrative nature, gliomas are often difficult ot surgically excise & become increasingly large mass lesions; they also destroy the brain tissue locally. Some primary brain tumors grow rapidly & are very lethal, despite aggressive therapy, whereas others are more slowly growing & have a relatively good prognosis if treated appropriately. Although primary brain tumors may be very malignant in the sense that they quickily lead to death, these tumors rarely metastasize. Histologic classification is very important in predicting growth rate, & thus prognosis, for primary brain tumors, most often arise from a lung or breast primary, destroy brain tissue locally, & are mass lesions.
|
|
|
Demyelinating Diseases
|
Demyelination = myelin is lost from the axon, but the axon remains intact (selective vulnerability). Characterized by the common histologic findings of loss of myelin & relative sparing of axons, & by a primary injury of the myelinating glial cell (oligodendrocyte-CNS, Schwann cell-PNS) or its myelin sheath. Several different pathogenetic mechanisms involved in this selective injury. Multiple sclerosis of the CNS & the Guillain-Barre syndrome (inflammatory demyelinating neuropathy) of the PNS are the classic immunologically mediated demyelinating diseases. The leukodystrophies are a group of demyelinating diseases: myelin loss is due to an inherited metabolic defect. The loss of the myelin sheath prevents the axon from effectively conducting nerve impulses, so that the axon loses its function. Fortunately, remyelination of axons by Schwann cells can occur in the PNS; these remyelinated fibers regain their ability to conduct impulses & permit return of function...There is no significant remyelination of axons in the CNS & no return of function.
|
|
|
Neurodegenerative Diseases
|
A group of disease in which the primary abnormality is a selective degeneration of certain populations of neurons. The clinical presentation of each disease is dictated by the which neuronal populations are involved. Thus, diseases characterized by degeneration of neurons of the cerebral cortex (i.e. Alzheimer's disease) are associated with dementia; diseases characterized by degeneration of the neurons of the basal ganglia are associated with movement disorders (i.e. Huntington's chorea); diseases characterized by degeneration of neurons of the substantia nigra (in brain stem) are associated with Parkinsonism (Parkinson's disease); & diseases characterized by degeneration of upper & lower motor neurons are associated with paralysis (e.g., amyotrophic lateral sclerosis). In some diseases, the neuronal degeneration is accompanied by characteristic histologic changes (e.g., neurofibrillary tangles, senile plaques, Lewy bodies). Most neurodegenerative diseases are sporadically; a few have Mendelian inheritance. The pathogenesis is not well known.
|
|
|
Amyotrophic Lateral Sclerosis (ALS; motor neuron disease)
|
-A neurodegenerative disease in which there is a preferential loss of the Upper & Lower Motor Neurons. Loss of upper motor neurons results in spastic paralysis, while loss of lower motor neurons results in flaccid paralysis & muscular atrophy
-Most cases are sporadic, but a small percentage of cases are familial. One mutated gene causing familial ALS encodes for superoxide dismutase (SOD1). The pathogenesis of the neuronal degeneration is unknown. -Histopathologically, there is loss of large motor neurons (Betz cells in motor cortex, motor neurons in lower brain stem, & anterior horn cells in spinal cord). Degeneration of the corticospinal tracts of the spinal cord occurs as a consequence of the loss of upper motor neurons. |
|
|
Acute Appendicitis
(Morphologic Findings) |
Gross --> congestion with a fibrinopurulent exudate on the serosal surface
Micro --> Mucosal ulceration with neutrophils infiltrating the wall of the appendix |
|
|
Endocrinology Definition
|
-study of the glands and the hormones they produce
-classically includes: pituitary, thyroid, parathyroid, adrenals, pancreatic islets, and gonads |
|
|
Hormones
|
-term derived from Greek "to set in motion"
-growth,maintenance of homeostasis,reproduction -some are synthesized as precursor polypeptides |
|
|
5 Classes of Hormones
|
-amino acid derivatives: catecholamines, thyroid hormones (T3/T4)
-small neuropeptides: thyrotropin -releasing hormone, somatostatin -large proteins: insulin, parathyroid hormone -steroid hormones: cortisol, estrogen, testosterone -Vitamin derivatives: vitamin A, Vitamin D |
|
|
Hormone and Protein Secretion
|
-peptide hormones initially stored in secretory granules and released after stimulation by releasing factor or neural signal
-steroid hormones are secreted as they are synthesized |
|
|
Hormone and Protein Binding Proteins
|
-Serum-binding proteins protect hormones from premature degradation
-restrict hormone access to some receptors and other sites -only unbound (FREE) hormone can interact with receptors and elicit a biologic response |
|
|
Endocrinology Definition
|
-study of the glands and the hormones they produce
-classically includes: pituitary, thyroid, parathyroid, adrenals, pancreatic islets, and gonads |
|
|
Hormones
|
-term derived from Greek "to set in motion"
-growth,maintenance of homeostasis,reproduction -some are synthesized as precursor polypeptides |
|
|
5 Classes of Hormones
|
-amino acid derivatives: catecholamines, thyroid hormones (T3/T4)
-small neuropeptides: thyrotropin -releasing hormone, somatostatin -large proteins: insulin, parathyroid hormone -steroid hormones: cortisol, estrogen, testosterone -Vitamin derivatives: vitamin A, Vitamin D |
|
|
Hormone and Protein Secretion
|
-peptide hormones initially stored in secretory granules and released after stimulation by releasing factor or neural signal
-steroid hormones are secreted as they are synthesized |
|
|
Hormone and Protein Binding Proteins
|
-Serum-binding proteins protect hormones from premature degradation
-restrict hormone access to some receptors and other sites -only unbound (FREE) hormone can interact with receptors and elicit a biologic response |
|
|
Hormone Regulation
|
-Feedback control: positive and negative
-negative feedback tends to dominate -positive feedback is less understood -other control factors: immune system, cytokines, & other local factors -maintain constant levels of hormones within a narrow range |
|
|
Biologic Rhythms
|
-Environmental adaptation
-seasonal changes -light-dark & sleep-wake cycles -meals -menstrual cycles -age circadian/diurnal patters |
|
|
Pathologic Mechanisms of Endocrine Disorders
|
-Hormone excess
-hormone deficiency -Hormone Resistance (at or post receptor) |
|
|
Hormone Excess
|
-neoplasms
-autoimmune -Iatrogenic -infectious/inflammatory -mutations at key receptors |
|
|
Hormone Deficiency
|
-autoimmune
-iatrogenic -infectious/inflammatory -nutritional/vitamin deficiency -defect/mutations -hemorrhage/infarction -hormone resistance |
|
|
Approach to Endocrine Patient
|
-symptoms reflect amount of available hormone
-hormone measurements in serum/plasma or urine (baseline samples collected under controlled conditions) * Deficiency = stimulation study *Excess=suppression study -primary disease -secondary disease |
|
|
Pituitary Gland
|
-1cm in length; .5grams
-below hypothalamus in bony sella turcica attached to hypothalamus by infundibulum -anterior lobe is composed of cords of secretory cells in righ vascular stroma -secretory cells named for staining properties: Acidophils (A) or Basophils (B) or named for secretory product |
|
|
Acidophils
|
-somatotrophs (GH)
-Lactotrophs (prolactin) |
|
|
Basophils
|
-Thyrotrophs (TSH)
-Corticotrophs (ACTH) -Gonadotrophs (FSH & LH) |
|
|
Releasing and inhibiting Factors
|
-CRF
-GnRH -GHRH -PIF -TRH -SRIF |
|
|
Tropic Hormones
|
-ACTH
-LH -FSH -GH -PRL -TSH |
|
|
Secretory Hormones
|
-cortisol
-testosterone -estradiol -IGF-1 -T4/T3 |
|
|
Anterior Pituitary Insufficiency
|
-may be acquired or developmental/genetic
-acquired is most common (tumors, inflammation, vascular damage) -hormone loss can be selective, partial, or complete -sequence of hormone los related to compression or destructive lesions: GH then LH, FSH then TSH finally ACTH -initial symptoms due to insufficient levels of target hormones -Adults-usually hypogonadism -children-growth retardation |
|
|
Anterior Pituitary Excess
|
-Primary disease
* pituitary adenomas, hyperplasia (prolactin secreting adenomas, GH or ACTH secreting tumors) -Secondary disease (damage to hypothalamus or hypothalmic - pituitary tract, decreasing inhibiting factors |
|
|
Growth Hormone
|
-Action: promotes growth of soft tissues, cartilage, and bone
-released in response to exercise,stress, hypoglycemia, and other hormones (testosterone, estrogen, thyroxine) -pulsatile secretion (peak concentrations during sleep, p meals) -random measurments not useful -GH stimulates IGF synthesis -Stimulation or suppression tests |
|
|
Growth Hormone and IGF-1
|
-excess: acromegaly (adults) , giantism (children)
*increased GH: multiple samples/24h, increased IGF-1: better correlation to clinical severity -Deficiency: decreased IGF-1 not specific, r/o nutrition, psychological, hepatic dysfunction, correlate with age, GH stimulation test |
|
|
Growth Hormone excess
|
-pituitary adenoma (giantism in children and acromegaly in adults)
-Features become more pronounced with time (nose, jaw, tongue{marcoglossia}, soft tissues of hands and feet become enlarged, overgrowth of mandible leads to malocclusion, teeth may become separated, generalized apical root resorption) -compression or destruction of normal pituitary tissue will lead to hypofunction of other systems (secondary hypothyroidism etc) -fatigue, osteoarthritis, osteoporosis |
|
|
Adrenal Glands
|
-pair ~4cm (each located at superior pole of kidney)
-cortex -medulla |
|
|
Cortex of Adrenal Glands
|
-hormones synthesized from cholesterol
-zona glomerulosa: mineralcorticoids (aldosterone: Na and K regulation_ -Zona fasciculata : Glucocorticoids (cortisol: protein, fat, carb metabolism, *responsive to ACTH) -Zona reticularis: Androgens (testosterone, dehydroepiandrosterone, androsterdeione-sexual maturation, *responsive to ACTH) |
|
|
Adrenal Medulla
|
-Hormones synthesized from tyrosine (Epi, NorEpi, Dopamine)
|
|
|
Cortisol
|
-regulated by ACTH and immune system
-~90% bound to cortisol-binding globulin (CBG) albumin and sex hormone binding globulin -exhibits circadian rhythm or pattern (highest in AM and lowest in PM) |
|
|
Adrenal insufficiency
|
-Primary-Addison's disease
-worldwide : infectious cause: TB, histoplasmosis, crytococcosis, CMV -US: autoimmune adenalitis, watch for other autoimmune diseases -gradual onset -generally affects entire cortex leading to deficiency in all adrenal sterioids -Secondary: decreased pituitary ACTH |
|
|
Addison's Disease
|
-glucocorticoid deficiency (weakness, weight loss, fatigue, hypoglycemia, nausea)
-Mineralocorticoid deficiency (hyponatremia, hyperkalemia, dehydration) -ACTH excess (hyperpigmentation of skin and mucus membranes{melanocyte stimulating hormone}) |
|
|
Crohns Disease
(Gross Pathology) |
-Discontinuous involvement of GI (can affect stomach, small intestine and large intestine)
-Transmural inflammation leads to thickening of wall (creeping fat= mesenteric fat wraps around affected area -fistulas -mucosal ulceration and "cobblestone" appearance |
|
|
Crohns Disease
(Microscopic Pathology) |
-active inflammation (cryptitis = intraepithelial neutrophils; occasional crypt abscess=neutrophils in crypt lumens)
-features of chronicity (crypt branching/drop-out -mucosal ulceration -transmural lymphoid aggregates -non-caseating granulomas |
|
|
Chrons Disease
(Local Complications) |
-small bowel obstruction (common) * due to stricture formation
-Iron deficiency (ulceration may cause occult hemorrhage, frank rectal bleeding can occur) -perforation (in 2% of cases) -fissuring and fistulation can lead to intra-abdominal abscess formation -malabsorption (if extensive small bowel involvement, vitamin B12) |
|
|
Ulcerative Coliti
(Gross Pathology) |
-continuous involvement (distal rectum to variable length proximal)
-involves colon and rectum only -inflammation limited to mucosa -pseudopolyps |
|
|
Pseudopolyps
|
-islands of intact, regenerative mucosa surrounded by adjacent ulcerated/inflamed mucosa
|
|
|
Ulcerative Colitis
(Microscopic Pathology) |
- Active inflammation (cryptitis =intraepithelial neutrophils; crypt abscess=neutrophils in crypt lumens)
-features of chronicity (crypt branching/drop-out) -mucosal ulceration |
|
|
IBD & Colon Cancer
|
-both UC and CD are associated with an increased risk of developing colon cancer
-Risk may be the result of persistent inflammation and repair -greater risk is associated with severe disease involving the entire colon for over 10 years and onset of disease in childhood |
|
|
Diverticular Disease
|
-protrusions of the mucosa through the muscle wall
-common in western societies -50% of ppl >60 yrs -low bulk diet -extremely uncommon in underdeveloped nations |
|
|
Diverticular Disease
(Gross & Microscopic findings) |
Gross:
-range in number (few to many) -arranged in parallel rows (usually sigmoid colon) -may contain impacted and calcified stool (fecalith) Micro: -flask-like outpouching of mucosa and submucosa through muscularis |
|
|
Diverticular disease: Complications
|
-asymptomatic in 80% of afected individuals
-inflammation (diverticulitis) resulting in perforation and pericolonic abscess, hemorrhage- erosion into major blood vessels present in the connective |
|
|
Adenomatous Polyps
|
-benign gland forming, epithelial neoplasms of colonic mucosa
-dysplasia: disturbance in growth and differentiation manifest as architectural and cytologic dysplasia -dysplasia can cary in degree: mild moderate or severe -adenomatious polyps are the precursor lesions of the vast majority of colorectal cancers -though not every adenoma will give rise to colorectal cancer -remain the same size for prolonged periods or even regress |
|
|
Adenomatous Polyp Growth Patterns
|
-sessile
-pedunculated |
|
|
Adenomatous Colonic Epithelium
|
-pleomorphic, hyperchromatic nuclei
-nuclear pseudostratification -loss of cytoplasmic mucin |
|
|
Colorectal Cancers
|
-most arise from pre-existing adenomas
-prevalenc of adenomas in pop. typically paralles the prevalenc of colorectal cancer -when invasive carcinoma is found at an early stage, there are usually adenomatous areas surrounding it -development of adenomas precedes that of colorectal cancer by 5-10 yrs -the risk of cancer is directly related to the number of adenomatous polyps -individuals with familial adenomatous polyposis syndrome are almost certain to develop colorectal cancer -removing adenomas decreases incidence of subsequent carcinoma |
|
|
Colorectal carcinoma
|
-annumally accounts for 150,000 new cases and 60,000 deaths in US
-Sporadic : 90 - 95% of cases -familial adenomatous polyposis syndrome |
|
|
Familial Adenomatious Polyposis syndrome
|
-1% of cases
-caused by mutations of adenomatous polposis coli (APC) gene on chromosome 5q21 -affected individuals develop 100s to 1000s of adenomatous polyps by late adolescence which will progeress to carcinoma if untreated (colectomy) -30-40% of cases result from new germline mutation and thus do not have a family history to justify aggressive screening |
|
|
Colorectal Carcinoma : herditary nonpolyposis colorectal carcinoma syndrome
|
-5-10% of colorectal carcinoma
-autosomal dominant familial syndrome -colonic malignancies often multiple and not associated with adenomas -associated with extraintestinal carcinoma, particularly the endometrium - |
|
|
Colorectal Carcinoma
(Gross & Microscopic Findings) |
Gross:
-polpoid, ulcerating or infiltrative appearance -infiltrative tumors often present with annular or "napkin ring" constrictions leading to bowel obstruction Micro: -well, moderately or poorly differentiated invasive glands (adenocarcinoma) -prognosis related to degree of differentiation (grade), depth of invasion and nodal involvement (stage) |
|
|
Colorectal Carcinomas
(Clinical features) |
-occult GI bleeding with gradual development of anemia
-occasionally blood loss is sufficient to yield bright red blood in stool -obstruction -perforation |
|
|
Adenomatous transformation to Colorectal cancer
|
-approximately 10 yrs
-broad window of opportunity for prevention of colorectal carcinoma by endoscopic removal of polyp |

