![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
11 Cards in this Set
- Front
- Back
|
What is the general formula for alkanes? |
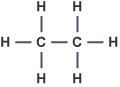
The general formula for alkanes is: Cn H2n+2. Ethane = C2 H6 |
|
|
What is the general formula for alkenes? |
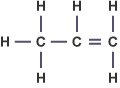
The general formula for alkenes is:
Example: Propene = C3H6 |
|
|
What is the general formula for alcohols? |
The general formula for alcohols is: Cn H(2n+1) OH.
|
|
|
What is the general formula for carboxylic acids? |
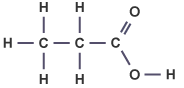
The general formula for carboxylic acids is: |
|
|
Explain the production of ethanol via fermentation. |
The raw material sugar (from sugar cane or sugar beet) is mixed with water and yeast at just above room temperature in a reactor vessel. The yeast contains an enzyme called zymase which acts as the biological catalyst to convert sugar to ethanol in fermentation, it works best at an optimum of pH ~4. Under anaerobic conditions at an optimum temperature of 30C to 40C, the sugars react via the enzymes in the yeast cells to form ethanol and carbon dioxide gas |
|
|
What is the word equation for the production of ethanol via fermentation? |
glucose (sugar) == enzyme ==> ethanol + carbon dioxide |
|
|
What is the symbol equation for the production of ethanol via fermentation? |
C6 H12 O6(aq) ==> 2C2 H5 OH(aq) + 2CO2(g) |
|
|
Explain how ethanol is separated from a fermented mixture? |
Ethanol is separated from the mixture by fractional distillation.! Ethanol has a lower boiling point (78C) than water (100C) and distils off first giving a concentration of up to 95% ethanol. The two vapours separate in the fractionating column with ethanol rising to the top, passing out into the condenser, condensing to a liquid for collection in a suitable container. Most of the water condenses back into a liquid in the fractionating column and runs back into the flask. |
|
|
Other than by fermentation, how else can ethanol be produced? |
Ethanol can also be produced by the reaction of steam and ethene (an alkene from oil cracking) in the presence of a strong acid catalyst (phosphoric acid, H3 P O4). |
|
|
What factors are required for the reaction between ethene and steam to take place? |
The reversible reaction is carried out at a moderately high temperature (e.g. 300C) and a high pressure (e.g. 60–70 times atmospheric pressure). |
|
|
In terms of the equilibrium, what would increasing the pressure do to ethene and steam? |
Increasing pressure shifts the reaction equilibrium to the right. |

